
INDIA PRIVATE LIMITED
Dear Valued Vedicinals family Members,
As we step into the New Year, the Vedicinals team extends heartfelt wishes for your health and a swift recovery in 2024. We sincerely appreciate the wealth of suggestions and feedback you’ve shared, and we want to express our gratitude for your trust and support in Vedicinals.
While perfection may be an ongoing pursuit, our commitment to doing our best remains unwavering. Your input is invaluable, and we eagerly welcome every suggestion and insight that aids us in our continuous quest for improvement.
In the spirit of growth and evolution, we encourage you to continue sharing your ideas, experiences, and suggestions. Your contributions empower us to enhance our services and cater to your needs more effectively.
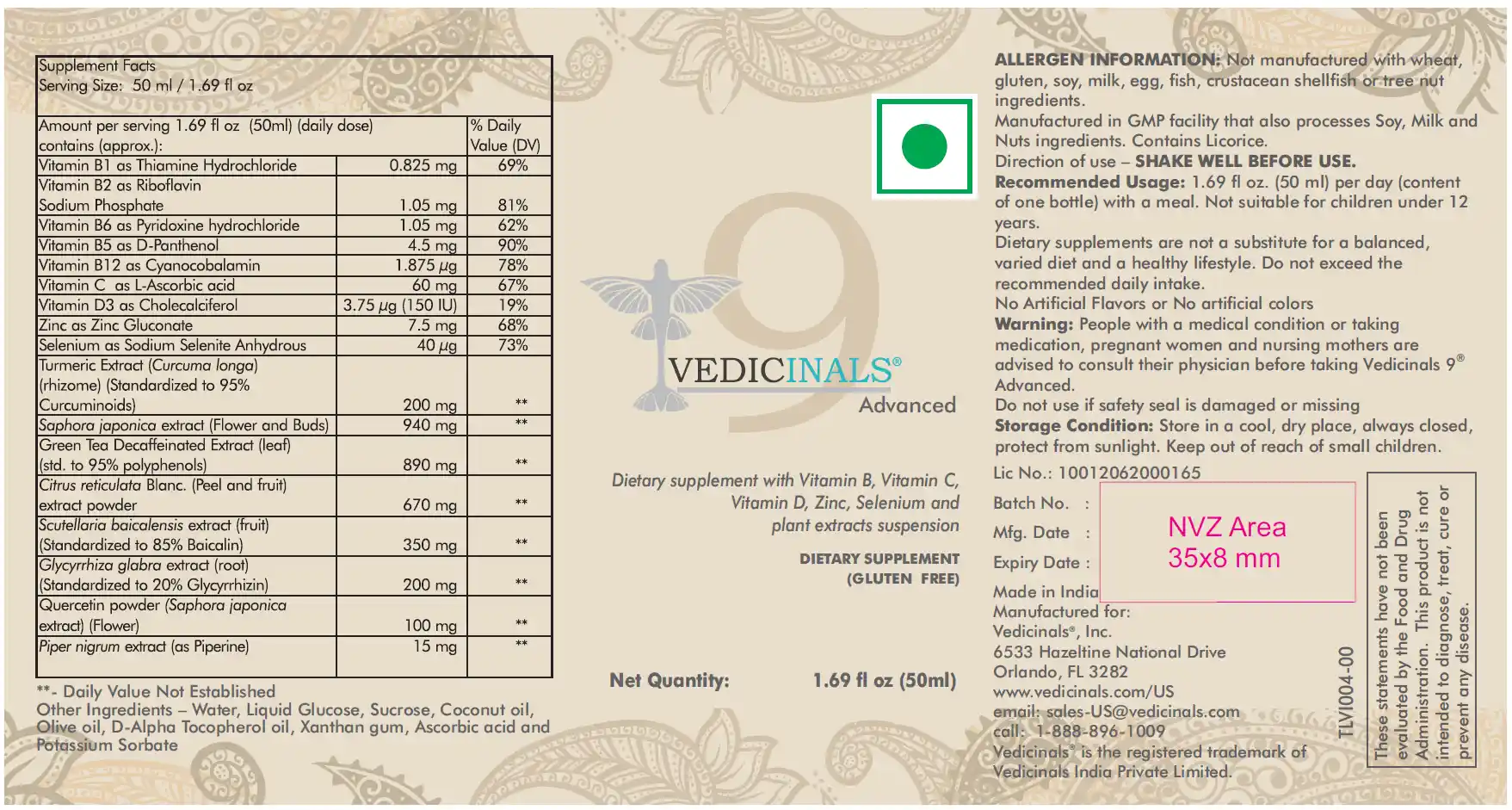
Now, let’s delve into the exciting updates for Vedicinals 9 Advanced:
1. Enriched Nutritional Support and Convenience:
In our continuous effort to enhance your health journey, we’ve revamped Vedicinals 9 Advanced to offer comprehensive nutritional support. By integrating 12 vital vitamins, minerals, and oils directly into our product, we’re simplifying your supplement regimen. Embrace the convenience of finding essential nutrients such as 1. Vitamin B1, 2. B2, 3. B6, 4. B5 (D-Panthenol), 5. B12, 6. C (L-Ascorbic acid), 7. D3, 8. E (D-Alpha Tocopherol oil), 9. Zinc, 10. Selenium, 11. Coconut oil, and 12. Olive oil all in one place. No longer will you need to purchase a multitude of supplements separately; our improved formula provides a holistic approach to meeting your nutritional needs with convenience.
2. Safest formulation in class:
Vedicinals 9 Advanced is designed for highest safety profile. Acute toxicity trial proved our formulation has achieved Category 5 , which is the highest category possible for Orally ingestible formulation. Our recommended daily dose is < 30 and < 60 mg/kg against the LD50 value > 5000 mg/kg.
3. Vegan, GMO Free , Gluten-free :
We’ve made notable updates: switching to vegan-friendly ingredients by replacing honey. Additionally, our product is now GMO-free and gluten-free, aligning with diverse dietary preferences and ensuring broader accessibility for all our users.
4. Improved Tolerance and Acceptability:
To enhance your experience, we’ve minimized excipients, additives, and preservatives, utilizing natural resources wherever possible. This adjustment aims to reduce tolerance issues some individuals were experiencing. Notably, we’ve switched the source of Luteolin from peanuts to Saphora japonica.
5. Compliance with Regional FDA Guidelines:
Ensuring your safety and satisfaction, Vedicinals 9 Advanced now fully complies with regional FDA guidelines. Our ingredients, additives, and labels adhere to US and EU FDA standards, complete with allergen warnings. Every ingredient undergoes rigorous screening for heavy metals and pesticide contamination. Our commitment to quality ensures that the final product not only complies but surpasses regulatory standards, guaranteeing top-notch safety and purity.
Our manufacturing facility is audited and approved by various FDA incl USFDA. Facility is NSF GMP certified with accreditations like ISO 14001, ISO 45001, OHSAS 18001, VEGAN….
6. Reduced Daily Dose:
Our committed research teams have achieved substantial progress in maximizing bioavailability and absorption within the human system. Consequently, we have successfully halved the daily dose compared to our previous product while preserving similar efficacy. This translates to your Vedicinals box now lasting twice as long.
We’re thrilled about these advancements, and we look forward to your continued support and feedback. Here’s to a healthier, happier 2024 with New Vedicinals!
Recommended Dosage for Vedicinals-9 Advanced : Half a Bottle ( 25 ml) Per day.
1 Box with 14 Bottles / Vials lasts 28 Days
This can be consumed in one time or divided into 2 doses.
Option 1 : Half a bottle after breakfast or
Option 2 : Quarter bottle after breakfast and a Quarter bottle after dinner.